The Bio Process Research and Development Laboratories builds.
#REATA RISK OVERSIGHT PROCESS PDF#
Personnel should follow guidance according to federal, state, local, tribal, and territorial regulatory requirements.įacilities should also perform a site-specific and activity-specific risk assessment pdf icon external icon to identify and mitigate risks. Since waste regulations vary from state to state, disposal must comply with all applicable local, regional, national, and international regulations.


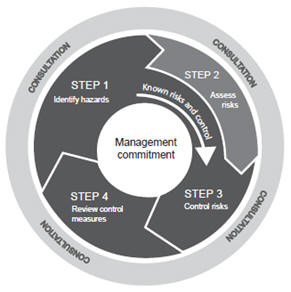
The PMA process is important for the regulation of commonly used dermatologic devices, many of which are Class III devices. When disposing of waste generated from SARS-CoV-2 point-of-care testing, laboratories and testing sites should treat all waste from suspected or confirmed COVID-19 patient specimens and kit components as biohazardous waste. While the PMA pathway was designed to ensure the safety of Class III devices, device modifications that are subject to less regulatory oversight may allow for postmarket introduction of risks to patient health. Recently, the importance of this responsibility has become more evident in the wake of an historic global financial crisis, which disclosed perceived risk management weaknesses across financial services and other organizations worldwide. Audience: Clinical Laboratory Professionals Risk oversight is a high priority on the agenda of most boards of directors.
